



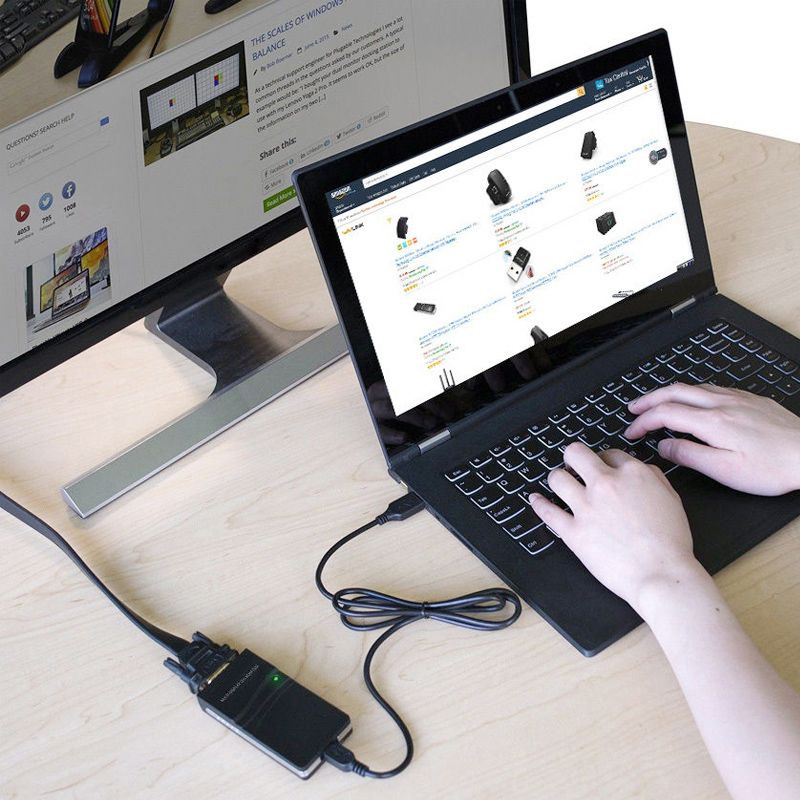
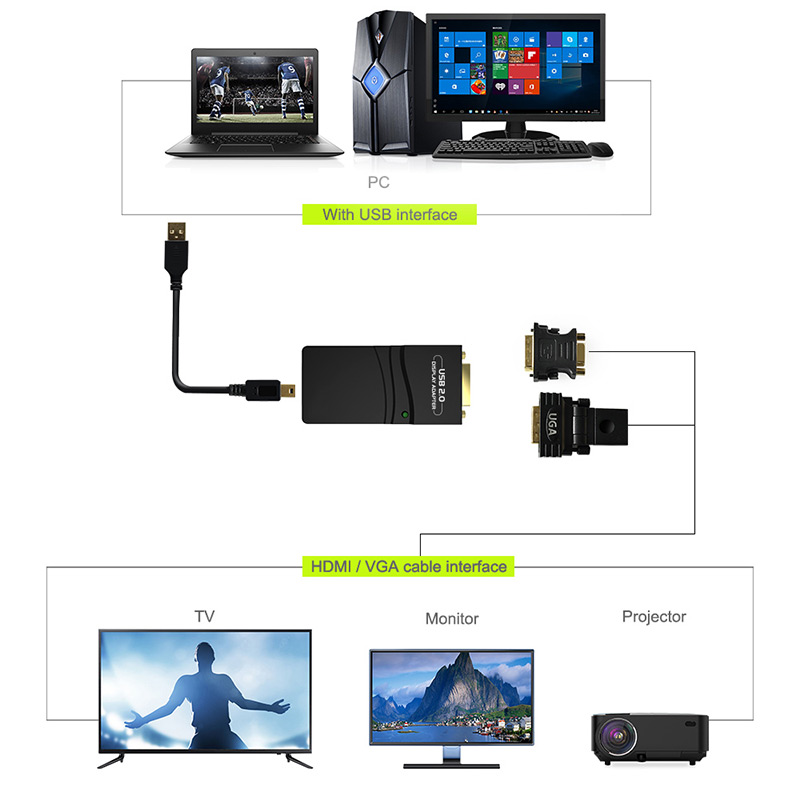
The adapter for multiple displays with mode extend. Just grab and go, the perfect travel companion and essential accessory for your trip around the world. Plug and play, maximum convenience.
WS-UG17D1
Data-driven insights allow for process optimization and reduced waste.
The traditional approach to process validation, once viewed as a one-time "three-batch" hurdle, has undergone a fundamental shift toward a science-based, lifecycle approach. This new paradigm, championed by the ISPE and regulatory bodies like the FDA, emphasizes that quality cannot be tested into a product; it must be built in by design. The Lifecycle Framework
The New Process Validation Paradigm represents a move toward a more "proactive" pharmaceutical quality system. By treating validation as an ongoing journey rather than a destination, the industry ensures a more reliable supply of safe, effective medicines for patients.
Early identification of variability prevents costly batch failures.
Perhaps the most critical shift, CPV requires ongoing monitoring during routine production. By using statistical process control (SPC), manufacturers can detect drifts or trends before they lead to failures, ensuring the process remains in a state of control throughout its entire commercial life. Why It Matters
It aligns with ICH Q8, Q9, and Q10 guidelines, facilitating smoother inspections and global compliance. Conclusion
This stage replaces the rigid "three-batch" rule. Instead, the number of batches is determined by risk assessment and statistical confidence. It confirms that the process design is capable of reproducible commercial manufacturing.
The focus here is on building a deep understanding of the product and process. By identifying Critical Quality Attributes (CQAs) and Critical Process Parameters (CPPs) early on, manufacturers can establish a "Design Space" where quality is guaranteed through scientific rationale rather than luck.